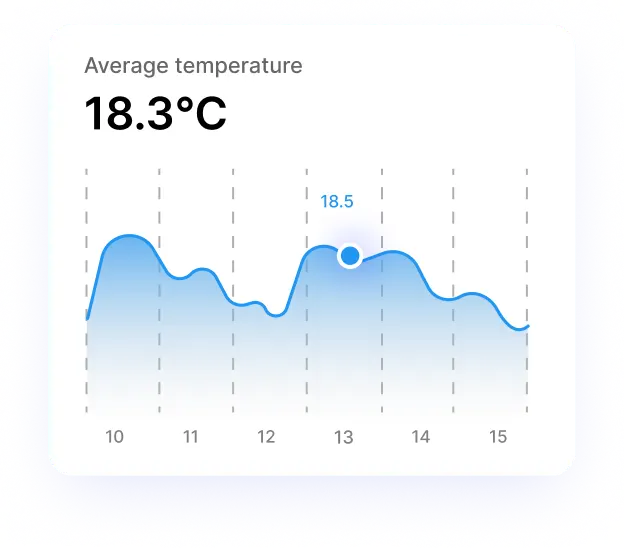
MonitoringMonitoring production environments to ensure pharmaceutical products meet quality standards
In the medical field, the ICG Datalogger integrates with medical devices to ensure continuous data monitoring, regulatory compliance, and quality assurance. Real-time data collection allows healthcare providers to optimize device performance and ensure patient safety.
Use case
Real-time monitoring of medical devices to track critical patient data, ensuring adherence to healthcare standards (e.g., ISO certifications) while providing predictive maintenance to prevent device failures.